Oncoral is novel daily irinotecan chemotherapy in development. Irinotecan chemotherapy has an established potent anti-tumor effect even in difficult to treat cancers. Oncoral is a daily irinotecan tablet with the potential to offer better patient outcomes with improved efficacy and safety following the daily dosing at home compared to intravenous high-dose infusions at the hospital. Following successful Phase 1 clinical studies, Oncoral is ready for Phase 2 clinical development.
ONCORAL IS A DAILY TABLET CHEMOTHERAPY FORMULATION
Oncoral is a patented oral tablet formulation of the topoisomerase I inhibitor irinotecan, a chemotherapeutic drug with a well established role and strong anti-tumor activity in the treatment of various cancer types.
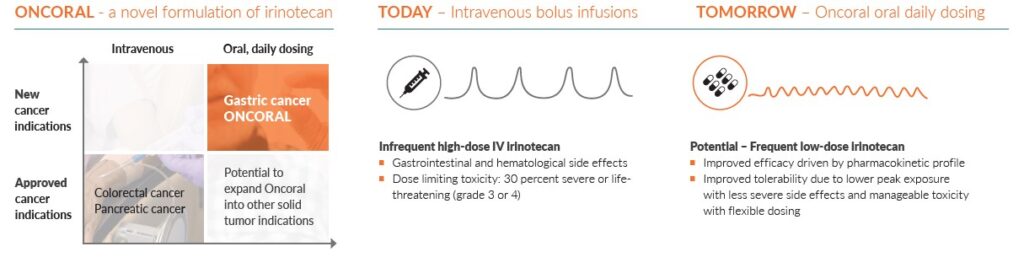
Oncoral is initially being developed for the treatment of gastric cancer in combination with other anti-cancer treatments.
The active substance in Oncoral is irinotecan, which has an established and proven effect in killing cancer cells. Irinotecan is a so called antineoplastic agent that after metabolic activation inhibits the enzyme topoisomerase 1, thereby inducing cancer cell death via the prevention of their DNA replication. Irinotecan is converted by carboxylesterases, primarily in the liver, to the active metabolite SN-38 which is 100-1,000 more potent than irinotecan in killing tumor cells. Data also shows a high conversion rate of irinotecan to the active metabolite SN-38 which has a high anti-tumor activity.
ONCORAL CAN BE THE FIRST ORAL VERSION OF IRINOTECAN
In Phase 1 clinical studies, the oral formulation has shown to enable a secure and efficient release and uptake of irinotecan from the gastro-intestinal tract. With oral administration, irinotecan can be given with low doses. This is a very different standard compared to giving a high intravenous dose every third week.
BENEFITS TO PATIENTS AND HEALTHCARE SYSTEMS
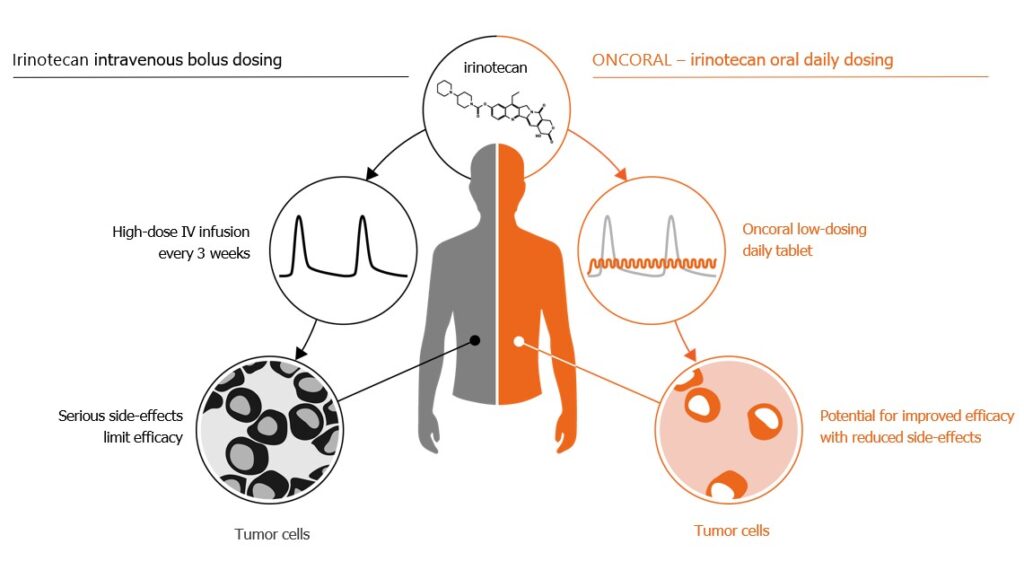
Treatment with currently available intravenous (IV) irinotecan infusions is often a trade-off between efficacy and tolerability for the patient, leading to sub-optimal outcomes for many cancer types, and toxicity related dose-reductions or discontinuations.
In addition, the suppressed immune function stemming from the chemotherapy treatment leads to an increased risk of infections and common gastrointestinal adverse events.
Oncoral is a daily irinotecan tablet with the potential to offer better patient outcomes with improved safety. Daily dosing can potentially improve efficacy driven by a more favorable, dosing-related, pharmacokinetic and pharmacodynamic profile. A continuous, low dose regimen also has the potential to reduce the severity of adverse events, infections, and other complications, including dosing flexibility and the possibility of faster discontinuation.
Furthermore, Oncoral has the potential to be combined with other chemotherapies and cancer drugs to potentially enable an all-oral combination option, which may reduce the burden of treatment and make adherence easier.
In addition, home administration can increase convenience for patients and alleviate healthcare staff and equipment allocated to hospital IV administration.
ONCORAL PHASE 1: ENCOURAGING RESULTS
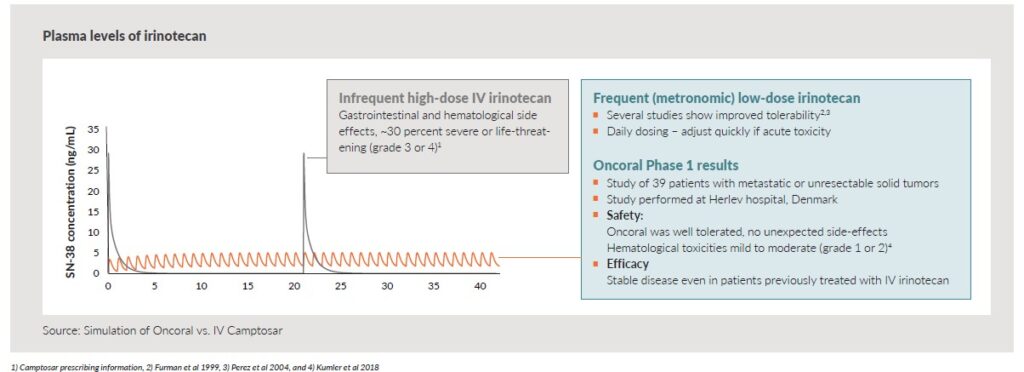
Oncoral has completed a Phase 1 trial at Herlev Hospital, Denmark. Data from the trial shows that Oncoral was well tolerated and confirmed the expected pharmacokinetics of Oncoral given as single agent. The study also established the maximum tolerated dose of Oncoral given as single agent and when administered in combination with another oral chemotherapeutic drug.
A total of 25 patients were enrolled in the first part of the study with Oncoral given as single agent. A further 14 patients were enrolled in the second part of the study where Oncoral was given in the combination study.
Oncoral Phase 1 studies were published in scientific journals:
Kümler et al. Cancer Chemother Pharmacol. 2019 Aug;84(2):441-446 (link)
Kümler et al. Cancer Chemother Pharmacol. 2019 Jan;83(1):169-178. (link)
| Phase 1 results – single agent study (publ. 2019) | Phase 1 results – combination study (publ. 2019) |
|---|---|
| Study: Dose escalating, open label, single center 25 patients with metastatic or unresectable solid tumors | Study: Open label, single center 14 patients with metastatic or unresectable solid tumors |
| Hematological toxicities were few and all mild (grade 1) to moderate (grade 2) | The combination of Oncoral with another oral chemotherapy, demonstrated reassuring tolerability |
| Pharmaco-Kinetic (PK) data showed consistent daily exposures during treatment at days 1 and 14 with no drug accumulation | |
| The active metabolite, SN-38, interpatient variability was in the same range as for IV administration |
ONCORAL HAS THE POTENTIAL TO IMPROVE BOTH EFFICACY AND SAFETY
Intravenous chemotherapy is often a trade-off between desired treatment effect and tolerability for the patient. With Oncoral as a daily irinotecan tablet there is a potential to improve both efficacy and tolerability compared to intravenous (IV) administration. In addition, it may offer convenience for the patient and at the same time reduce hospital costs with home administration.
Efficacy
The potential to improve efficacy is based on a fivefold higher conversion rate of irinotecan to the cytotoxic active metabolite SN-38 when dosed orally compared to an IV infusion. In addition, the principle of frequent, low daily dosing, also called metronomic dosing, may optimize the exposure of SN-38 and maximize the anti-tumor effect. Several studies provide proof of concept for metronomic dosing, including improved patient outcomes.
Safety
Conventional IV bolus administration of irinotecan is associated with toxicity. Most patients experience gastrointestinal and hematological side effects, of which approximately 30 per-cent are severe or life-threatening (grade 3 or 4, ref: Camptosar® prescribing information). Frequent low dosing, avoiding high peak plasma levels, may reduce toxicity and complications compared to high-dose IV infusions. Oral daily administration also brings the opportunity to adjust dosing quickly in case of acute toxicity.
IMPROVING IRINOTECAN EFFICACY AND TOLERABILITY
CLINICAL DEVELOPMENT STRATEGY AND PREPARING FOR PHASE 2 STUDY
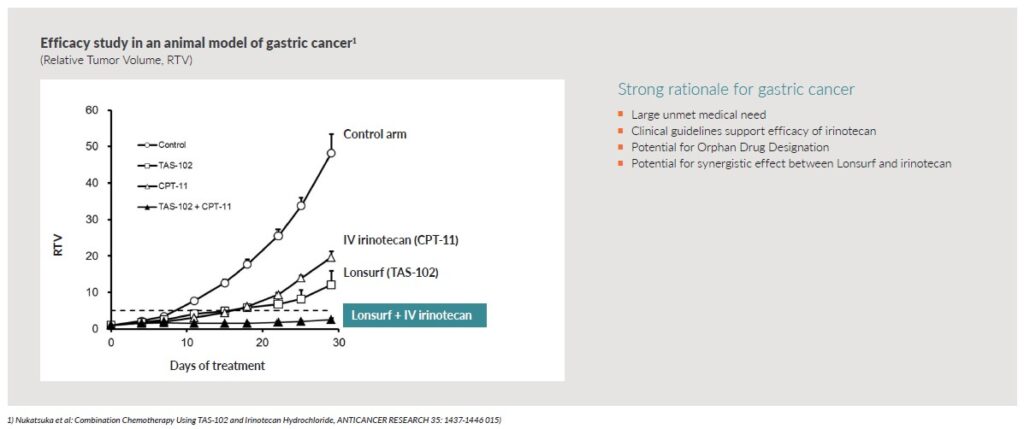
Potential for synergistic effect – The planned Phase 2 study will address metastatic gastric cancer. In the study, Oncoral will be combined with Taiho Oncology’s oral drug Lonsurf® that is used today for treating metastatic gastric cancer. The combination of irinotecan (the active substance in Oncoral) and Lonsurf has been tested in animal models, which showed that the combination almost stopped the tumor from growing and gave better results than administering them as monotherapies.
PHASE 2 STUDY DESIGN AND COLLABORATION

Please note that Oncoral is an investigational medicinal product and is not yet approved for use by regulatory authorities in any jurisdiction.